What Are The Possible Orbitals For N 3. What are the possible values of n and ml for an electron in a 4d orbital? To calculate the maximum number of electrons in each energy level, the formula 2n 2 can be used, where n is the principal energy level (first quantum number).
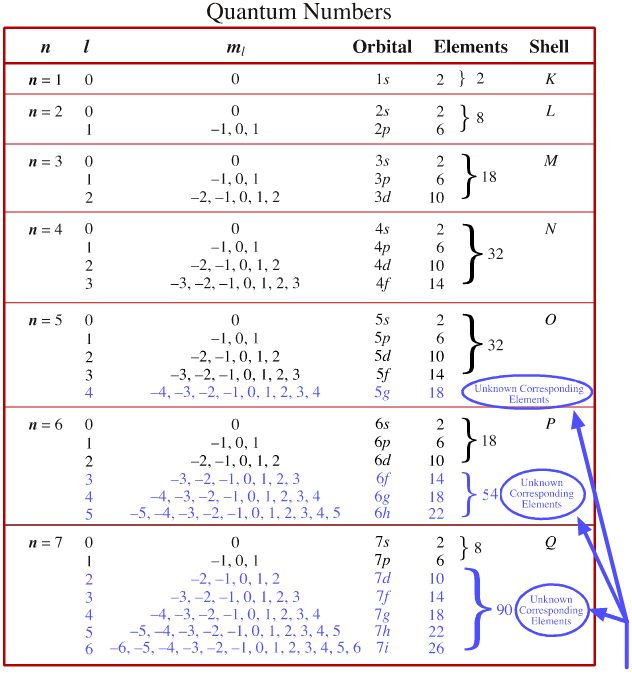
For n = 3 there are nine orbitals, for n = 4 there are 16 orbitals, for n = 5 there are 52 = 25 orbitals, and so on. Remember, here 0, 1, 2 doesn't contain the value of sum. The 3s, 3p, and 3d orbitals.
The Third Principal Power Degree Has Three Sublevels, S,P And D.the Sublevels Have Varied Numbers Of Orbitals, Which Are Areas Of Likelihood Of Discovering An Electron, And Every Orbital Can Have A Most Of Two Electrons.
In the 3rd energy level holds three subshells: In the 3s there is one orbital; Equally, it’s requested, how many orbitals are in the n 3 degree?
In The N=1 Shell You Only Find S Orbitals, In The N=2 Shell, You Have S And P Orbitals, In The N=3 Shell, You Have S, P And D Orbitals And In The N=4 Up Shells You Find All Four Types Of Orbitals.
It can also be noted that it is not possible for the 3f orbital to exist because that would require the value of ‘n’ and ‘l’ both to be equal to 3, which is not possible since the value of the azimuthal quantum number must always be lower than that of the principal quantum number. Within each shell of an atom there are some combinations of orbitals. So, in your case, if n is equal to 3, the values l must take are 0, 1, and 2.
∴ For L Is Equal To 1, The Minimum Value Of N Is 2.
When n = 1, l= 0 (l take away on one value and also thus there deserve to only be one subshell) as soon as n = 2, l= 0, 1 (l take away on 2 values and also thus there space two possible subshells) when n = 3, l= 0, 1, 2 (l takes on three values. 1p and 3f cannot exist. 11 how many orbitals can be there in l 3 subshell?
For L = 3, The Minimum Value Of N.
P, f sp os osp, d, f o s, p, d. 10 what is the maximum number of electrons that can have the quantum numbers n 3 l 2 ml 1? 16 orbitals for n = 3 there are nine orbitals, for n = 4 there are 16 orbitals, for n = 5 there are 52 = 25 orbitals, and so on.
3S, Which Carries One Orbital.
Corresponds to s, p , d, f orbitals respectively. To calculate the maximum number of electrons in each energy level, the formula 2n 2 can be used, where n is the principal energy level (first quantum number). We review their content and use your feedback to keep the quality high.